Severe eczema drug is approved by FDA; Price tag is US$37,000 a year
NEW YORK — The Food and Drug Administration on Tuesday (March 28) approved an expensive drug to treat people with a serious form of eczema, a potential breakthrough for people who have suffered for years without relief. But it will not come cheap.
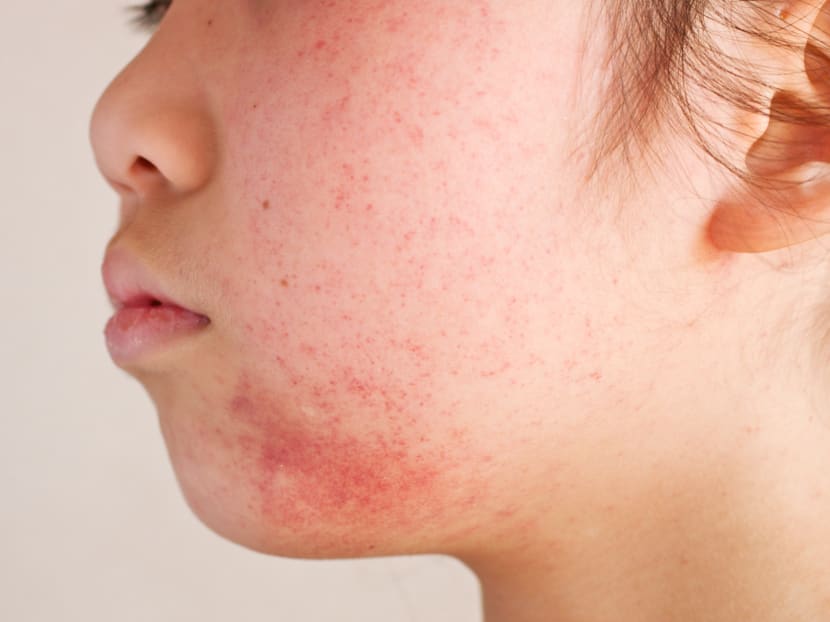
NEW YORK — The Food and Drug Administration on Tuesday (March 28) approved an expensive drug to treat people with a serious form of eczema, a potential breakthrough for people who have suffered for years without relief. But it will not come cheap.
The drug, to be called Dupixent, will carry a list price of US$37,000 (S$51,720) a year, a hefty price tag for patients who are increasingly being asked to pay a larger share of the drugs they take. Still, its price is a bit lower than many other commonly used biologic drugs, such as Humira and Enbrel, that treat other skin diseases.
In an effort to head off another public battle over the soaring cost of some prescriptions, the drugmakers Regeneron Pharmaceuticals and Sanofi took the unusual step of directly negotiating with insurers over the price and other details ahead of time. What consumers pay for drugs has come under heightened scrutiny in the last few years, most recently by President Donald Trump, who has promised several times to take drug companies to task over their costly products.
“Things have really deteriorated in our industry,” said Dr Leonard S Schleifer, the chief executive of Regeneron. “There’s so much concern over drug pricing that there’s an enormous amount of finger-pointing that is going on that’s counterproductive. It isn’t necessarily incentivising the right behavior.”
The best-selling products Humira and Enbrel, which treat the skin disease psoriasis and other conditions, carry annual list prices of about US$50,000.
Although many patients with insurance will not be asked to pay the full US$37,000, consumers have been more exposed to rising prices in recent years because insurers often require them to pay a percentage of a drug’s list price — sometimes up to half — or pay the full price until their deductible is met.
They also often must demonstrate that other, cheaper drugs did not work before an insurer will cover more expensive drugs. Regeneron has said it also negotiated a more streamlined approval process, although patients will still have to try other drugs first. It also said it had a patient assistance program to help people who have troubling paying.
Regeneron and Sanofi held much of the bargaining power in negotiating Dupixent’s price because it has been shown to work well and has no real competition, said Dr Steve Miller, the chief medical officer at Express Scripts, the nation’s largest pharmacy benefit manager, which often negotiates with drugmakers on behalf of clients like insurers and large employers.
With breakthrough treatments, “companies can really go to the high end of that demand for reimbursement”, Dr Miller said, “and all we have is the bully pulpit to complain about it”.
In trying to reach an agreement before approval, Dr Miller said, “This is how the system is supposed to work.”
Ms Christine Cramer, a spokeswoman for CVS Health, another leading pharmacy benefit manager, said, “While we believe our advocacy on behalf of our clients did ultimately influence Regeneron’s initial pricing strategy, the drug will be expensive”.
But Mr Peter Maybarduk, director of the Access to Medicines program at Public Citizen, a consumer group, noted that negotiations still took place out of the public eye and within the context of a system that is inherently unfair. “It’s the arrangement that corporations found beneficial in a system that provides monopolies and rewards secrecy, and we should ask for considerably more as the standard,” he said.
Dupixent treats severe to moderate atopic dermatitis, a common form of eczema that goes beyond the occasional bouts of itchy, dry skin that many people get. For people with serious forms of the disease, other treatments often do little to calm their skin, leading to sleeplessness, depression and social anxiety. Regeneron said on Tuesday that an estimated 300,000 people in the United States could qualify for its drug.
“I always say that atopic dermatitis doesn’t kill you, it just ruins your life,” said Dr Elaine Siegfried, a professor of pediatrics and dermatology at the St. Louis University School of Medicine. She was not involved in the clinical trials that led to approval, but she said she was likely to enroll patients in pediatric studies that are getting underway. Dupixent, she said, appears to work well, with few serious side effects. “It is groundbreaking,” she said.
Ms Debbie Byrnes, a sixth-grade teacher from San Antonio who participated in the clinical trial, had suffered for years with severe eczema that often covered her face. “That was the really difficult thing for me * I could never hide it,” she said. “I would have days when I went into school, and the kids would look at me and say, *What happened to you?’”
Ms Byrnes said she began noticing a difference about five days after her first dose and has now been using the drug for two years. Her skin is now almost completely clear, and occasional flare-ups are quickly brought under control. “If you saw me,” she said, “you wouldn’t know that I ever had atopic dermatitis.”
Before a drug is approved, pharmaceutical companies usually keep the price a secret and do not directly negotiate with insurers until the product is approved by the FDA, in part because of concerns that doing so could violate agency rules. But in January, the agency said that drug companies were allowed to discuss price and other issues with insurers in advance.
Even if Dupixent’s US$37,000 list price is below those of established drugs like Humira, Regeneron and Sanofi will very likely get to keep more than AbbVie, which makes Humira. That is because the net price of Dupixent, Dr Schleifer said, will still be over US$30,000. Although the rebates that drugmakers negotiate with insurers are not public, Mr Richard T Evans, an analyst for SSR Health, a stock analysis company, has estimated that AbbVie would get to keep less than that, or about US$27,500 a year for each patient who uses Humira.
He said a similar drug, nemolizumab, is also being tested for people with moderate to severe eczema and could hit the market in a few years. “Right now they have the benefit of being alone,” Mr Evans said. “But going forward, I think there will be a lot of price competition.”
In reaching tentative agreements ahead of time, Regeneron and Sanofi appear to have been trying to avoid the pushback they encountered in 2015, when their expensive drug to treat high cholesterol, Praluent, was approved. Although Praluent and a similar drug, known as PCSK9 inhibitors, have been found to be beneficial to patients who don’t respond to other treatments, insurers have limited access and sales have not lived up to expectations.
“We all got that wrong,” Dr Schleifer said. “But if we can make all of it work so that everybody’s feeling good, then we’ve really changed the paradigm.” THE NEW YORK TIMES