Doctors in S’pore who gave vaccine linked to South Korean deaths say no patients have reported complications
SINGAPORE — Several clinics in Singapore where doctors administered an influenza vaccine linked to deaths in South Korea said no patients have reported complications. They stopped using the vaccine after following an advisory from the Ministry of Health (MOH), which also advised temporarily ceasing use of a second vaccine.
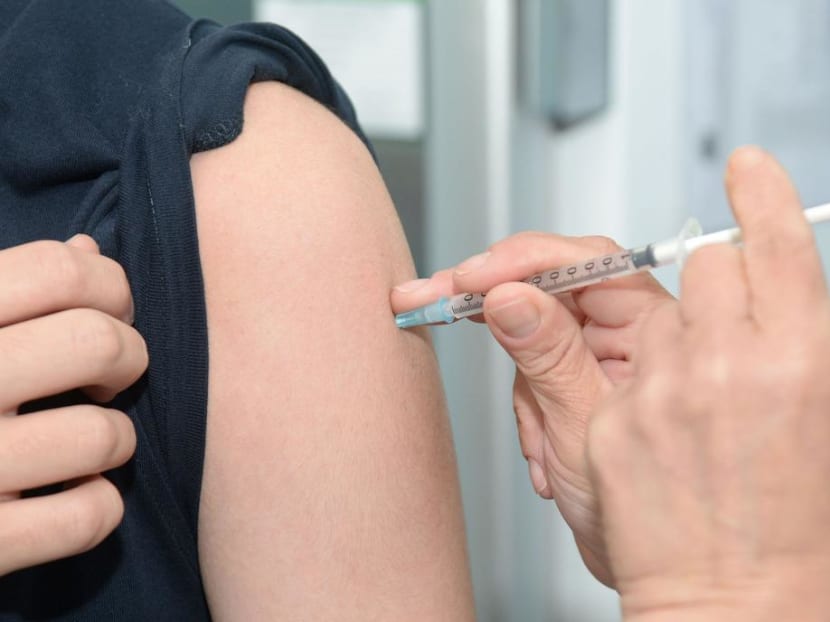
MOH advised on Oct 25, 2020 that medical practitioners should temporarily cease using two vaccines linked to deaths in South Korea.
- On Sunday, MOH advised medical practitioners here to temporarily cease using the two influenza vaccines
- The vaccines had been given to patients in South Korea who later died
- TODAY spoke to 14 doctors here, of whom six had used one of the two vaccines
- No patients who received the vaccine had reported complications, they said
SINGAPORE — Several clinics in Singapore where doctors administered an influenza vaccine linked to deaths in South Korea said no patients have reported complications. They stopped using the vaccine following an advisory from the Ministry of Health (MOH), which also advised temporarily ceasing use of a second vaccine.
While some doctors have checked in on their patients or received inquiries from those concerned about possible complications, others are waiting for more information from MOH, various clinics told TODAY on Tuesday (Oct 27).
MOH issued an advisory on Sunday to healthcare providers and medical practitioners to temporarily stop the use of SkyCellflu Quadrivalent, manufactured by SK Bioscience and locally distributed by AJ Biologics, and VaxigripTetra, manufactured by Sanofi Pasteur and locally distributed by Sanofi Aventis.
According to the South Korean health authorities, these two brands of vaccines, along with five others not available in Singapore, were administered to individuals who died there.
Eight out of 14 doctors with whom TODAY spoke said that they do not use either of the vaccines.
Six of them said that they had administered Vaxigrip Tetra previously — although only three had done so recently. No patients have reported side effects from the vaccine. None of the 14 doctors has used SkyCellflu Quadrivalent.
Dr Ong Eng Keow from the International Child and Adolescent Clinic at Mount Alvernia Hospital said that the clinic had administered about 50 Vaxigrip Tetra vaccines since Oct 4.
“We contacted all our patients to check on their condition and they are all well,” he said on Monday, adding that he had been most concerned about patients who got the jab recently.
Dr Choo Kay Wee, from A Life Clinic at Novena Medical Centre, has administered fewer than 10 Vaxigrip Tetra vaccines in the last six months.
Dr Sunil Kumar Joseph, who runs Tayka Medical Family Clinic in Jurong, said he had also administered fewer than 10 shots of the vaccine in the first half of this year, but none since then.
Neither doctor has seen patients coming back with complications.
Dr Joseph said he has not reached out to patients to inform them about the MOH temporary cessation advisory of the two vaccines as he is still waiting for more information from MOH.
“MOH is investigating the situation to see whether this is really warranted, or were there other issues that were not linked to the vaccine that may have caused those deaths (in Korea),” Dr Joseph added. “Let people investigate first before we alarm the patients.”
Some doctors said they have received inquiries from concerned patients since Monday.
Dr Lim Kien Sin from Clarion Medical Clinic in Ang Mo Kio said that the clinic received inquiries from patients even though it only started vaccinating last week — using different brands — and does not use either of the two vaccines that are the subject of the MOH advisory.
"The same few patients that we vaccinated gave us a call. We reassured them, and they were still quite all right," said Dr Lim.
TODAY contacted two private hospitals, Farrer Park Hospital and Thomson Medical Centre, which both said that they do not administer the two vaccines.
COMMON SIDE EFFECTS INCLUDE SWELLING, MILD PAIN
The doctors said there are several common side effects from vaccinations, such as mild pain at the injection site, swelling, rashes, mild body aches and fatigue.
Dr Lim said patients may also develop mild cough and cold symptoms, but this reaction usually resolves itself quickly.
“Most importantly, we look out for fever within the first 24 to 48 hours,” he said, adding that this is another common side effect of vaccinations.
Some patients may develop allergic reactions, the doctors said.
Dr Elaine Chua from Bedok Medical Centre said side effects such as rashes must be assessed in case the recipient of the vaccine has allergies to it.
Dr Lee Joon Loong from Paddington Medical Clinic in Bedok said that in severe cases, allergies can result in an anaphylactic shock — where their blood pressure suddenly drops and airways narrow, possibly blocking normal breathing.
In some cases, patients may feel numbness in their hands and feet after they are vaccinated, which can progressively move to more limbs, said Dr Chua.
This rare complication is called the Guillain-Barre syndrome, said Mount Alvernia’s Dr Ong, adding that patients may feel a weakness of muscles in response to influenza vaccines.
Referring to the various side effects that might be expected following a vaccination, Dr Chua said pain that is made tolerable with mild over-the-counter painkillers, such as pain on the injection site, is acceptable.
However, patients who develop more painful symptoms must be assessed by a doctor.
“Apart from the common side effects, the healthcare provider who administered the vaccine should be alerted to everything else,” said Dr Chua. She added that the healthcare provider must examine the patient to determine whether the side effect will progress and, if so, alert the Health Sciences Authority.
TODAY has sought comment from MOH.