New Singapore-developed test kit can tell within an hour if someone has had Covid-19
SINGAPORE — A new Covid-19 test kit invented by a research team at Duke-NUS Medical School can detect within an hour, instead of several days, if an individual has been infected before.
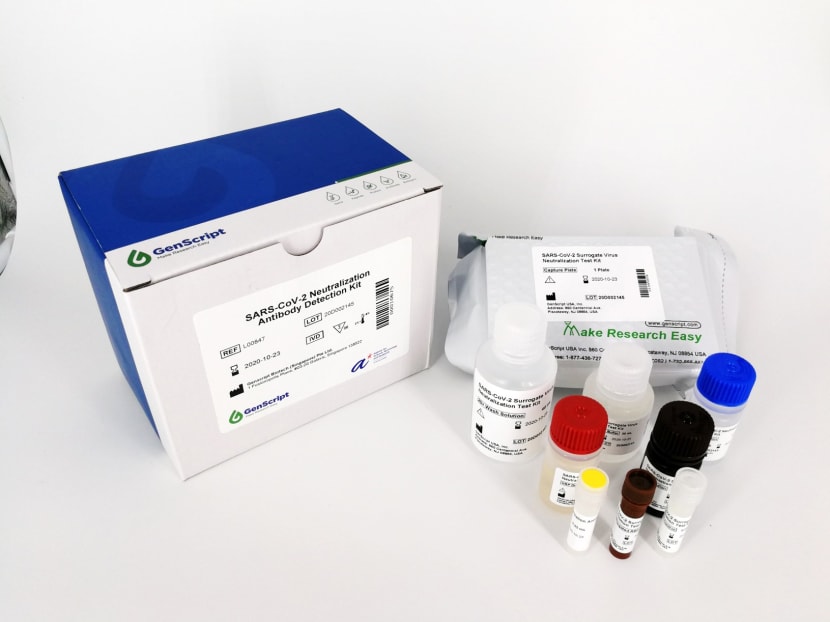
The cPass test kit produces results that are useful for facilitating contact tracing, developing potential vaccines and assessing whether a recovered patient is safe to return to work.
SINGAPORE — A new Covid-19 test kit invented by a research team at Duke-NUS Medical School can detect within an hour, instead of several days, if an individual has been infected before.
Called the cPass, the test kit produces results that are especially useful for facilitating contact tracing, developing potential vaccines and assessing whether a recovered patient is safe to return to work.
It was unveiled in a virtual press conference on Friday (May 15) by the medical school, biotechnology company GenScript Biotech Corporation and Singapore’s Agency for Science, Technology and Research (A*Star).
The kit is now available to Singapore hospitals.
The cPass works by examining blood samples to detect antibodies specifically created by the human body to fight Covid-19.
Antibodies are molecules produced by the body’s immune system to battle pathogens. If a person has recovered or is recovering from an illness, their body will still contain the antibodies that were spawned to resist it.
One such test was previously used to establish links between the Grace Assembly of God and Life Church and Missions Singapore Covid-19 clusters even while those tested had not shown any symptoms at the time.
Unlike other such tests, cPass can also be used in most research and clinical labs because it does not require a live sample of the coronavirus, removing the need for special facilities, highly skilled operators and complex lab procedures.
Director of Duke-NUS’s emerging infectious diseases programme Professor Wang Linfa, who led the team that invented cPass, said this makes the new test kit cheaper than other tests. He and representatives of GenScript and A*STAR, however, did not reveal the price of the test kit.
“It does not require a biosafety containment facility, which makes it immediately accessible to the global community, including many developed nations,” he said.
He stressed that the cPass is not meant to replace the polymerase chain reaction (PCR), the method Singapore currently uses to test for Covid-19. It detects the presence of the coronavirus in a swab sample and identifies active infections.
Instead, the cPass is meant to complement the PCR method which is unable to identify previous infections already cleared from the patient’s body.
The cPass can also be used to test animals, providing authorities with an advantage when tracking down the origin of the coronavirus which is widely believed to have come from infected animals.
Prof Wang said that work on the new test kit began on March 10 and that the cPass received provisional authorisation from the Health Sciences Authority on May 8.
Global biotech company GenScript helped with the proof concept research, product design and development, and optimisation and will help to commercialise the cPass.
A*STAR’s Diagnostics Development Hub will produce the pilot batch for use in Singapore hospitals. It had also developed the manufacturing protocol and quality controls for cPass’s provisional authorisation.
The joint team plans to work with local biotech companies to scale up production of the test kit.