Singapore studying effectiveness and safety of Moderna, Pfizer-BioNTech’s Covid-19 vaccines for children 6 months and older
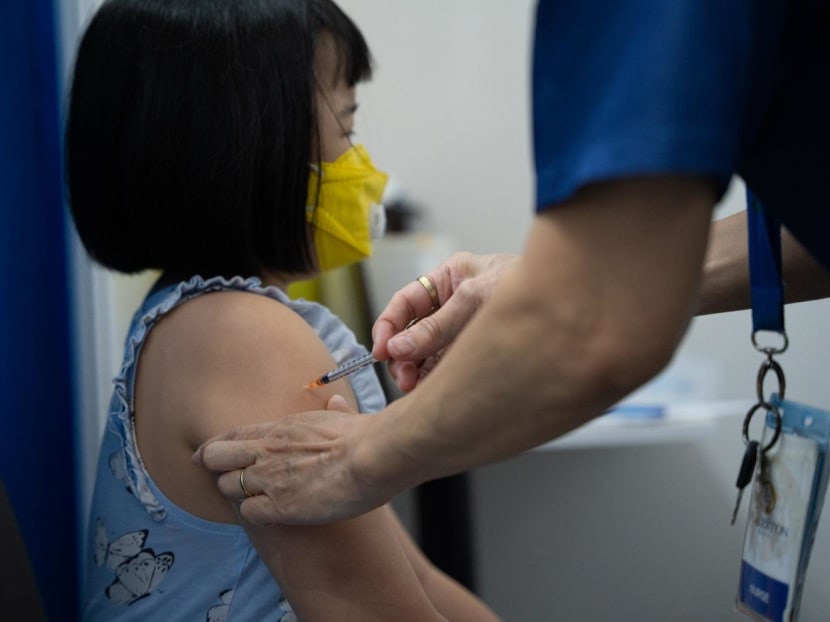
SINGAPORE — The Ministry of Health (MOH) and the expert committee on Covid-19 vaccination are now studying the effectiveness and safety of Moderna's and Pfizer-BioNTech’s vaccines in young children.
The study will look at the use of Moderna's Covid-19 vaccine for children aged six months to five years, and Pfizer-BioNTech's vaccine for those aged six months to four years.
The vaccine from both bio-pharmaceutical brands uses messenger ribonucleic acid (mRNA) to set up an immune system response to fight infection rather than part of an inactivated virus.
MOH said on Monday (June 20) in response to media queries: “We are engaging the respective vaccine manufacturers to obtain the relevant information to facilitate a risk-benefit evaluation of the use of these vaccines in this age group.”
The US Centers for Disease Control and Prevention last Saturday recommended Covid-19 vaccines for children as young as six months, allowing a nationwide rollout to start next week. The US CDC's move came after a panel of advisers voted to recommend Covid-19 vaccines for this group of children.
The US Food and Drug Administration last Friday authorised Moderna's vaccine for children aged six months to five years, and Pfizer-BioNTech's vaccine for children aged six months to four years. Pfizer-BioNTech's vaccine is already authorised for children over the age of five in the US. CNA
For more reports like this, visit cna.asia.